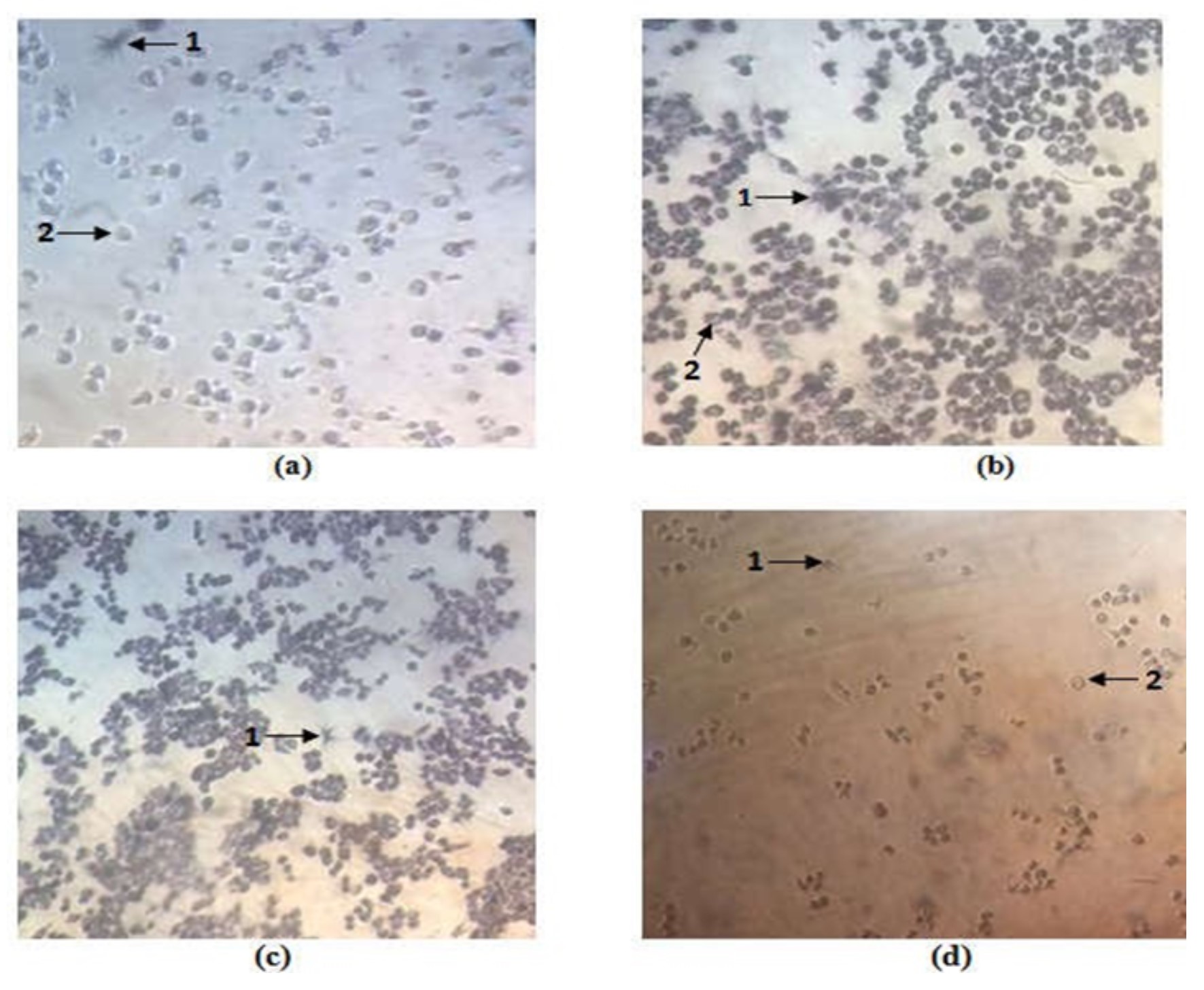
The Effect Cytotoxic and Apoptotic Propagation Chloroform Fraction of Ocimum sanctum L. Towards Hela Cells Line Culture
Abstract
Materials used in the research are Ocimum sanctum L, a herbaceous shrub used in traditional medicine displaying as immunomodulator, anti-stress, hepatoprotective, kemopreventif, and anti-inflammatory. A research to identify its ethanol extract of Ocimum sanctum has been conducted, showing that it possesses cytotoxic activities to HeLa cells with IC50 value of 209µg/ml. The aim of this research is to find out activities cytotoxic and apoptotic propagation chloroform fraction of Ocimum sanctum towards HeLa cells line. The extraction was done using maceration using ethanol 96% solvent and fractionation was done using chloroform. Effects of chloroform fraction on HeLa cells were tested using MTT at various dosages (500, 250, 125, 62.5, and 31.25) µg/ml. Cisplatin (Kalbe) was applied as positive control. The effect apoptotic propagation chloroform fraction of Ocimum sanctum on HeLa cells was observed by DNA painting method using orange ethridium bromide-acridine. The test results showed that the chloroform fraction of Ocimum sanctum extract on HeLa cells had an IC50 value of 155.67 µg / ml ± 46.99 and Cisplatin had an IC50 value of 16.81 µg / ml ± 20.37. Double staining test shows the existence of cells undergoing apoptosis. Based on these results, the chloroform fraction Ocimum sanctum has been shown to be cytotoxic and has the possibility of stimulating apoptosis of cervical cancer cells (HeLa). This can be used as a basis for the development of these plants as anticancer agents with specific action targets.
References
Christina, O. (2019). Citotoxic Activities And Expression of Genes P53 And Bcl-2 Extract And Fraction Of Basil Leaf (Ocimum sanctum L.) To T47d Breast Cancer Cells. Thesis, Faculty of Pharmacy, Setia Budi University of Surakarta
Haryanti, S. & Katno. (2011). Cytotoxic activity of Ocimum sanctum L On Cancer Cells Colon Widr. Surakarta: Simposium Nasional XV. PERHIPBA.
Ismiyati, N & Nurhaeni, F. (2016). The Effect Of Ocimum sanctum L. Leaves Ethanolic Extract As A Chemopreventive Agent In Hela Cervic Cancer Cells Through Cytotoxic Activity And Induced Apoptosis. Media Farmasi; Vol.3: 35-48.
Kurniandari, N., Susantiningsih, T., dan Berawi, K.N. (2015). The Effect of Lime Peel Extract (Citrus aurantifolia) as Nephroprotector to Histopathological Kidney Induced by Cisplatin. Majority; Vol. 4(9):140-143.
Magesh, V., Lee, J.C., Ahn, K.S., Lee, H.J., Lee, E.O., Shim, B.S., Jung, H.J., Kim, J.S.., Kim, D.K., Choi, S.H., & Kim, S.H. (2009). Ocimum sanctum Induces Apoptosis in A549 Lung Cancer Cells and Suppresses the In Vivo Growth of Lewis Lung Carcinoma Cells. Phytother Res; 23(10): 1385-1391.
Nissa, C.K., Oehadin, A., Martakusumah, A.H., Dewi, Y.A. (2015). Accuracy Comparison of Various Formulas for Estimating Glomerular Filtration Rate in Advanced Nasopharyngeal Carcinoma Patients before Cisplatin Administration. Majalah Kedokteran Bandung; Vol.47 (1).
Prasad, S., Yadav, V.R., Aggarwal, B.B. (2011). Ursolic Acid, a Pentacyclic Triterpen, Potentiates TRAIL-Induced Apoptosis through p-53 independent Regulation of Death Receptor : Evidence for The Role of Reactive Oxygen Spesies and JNK, J. Biol. Chem., 286(7) : 5546-5557
Rasjidi, I (2009). Early Detection & Prevention Of Cancer In Women. Jakarta: Sagung Seto.
Sekti, D.A., Muhammad, F.M., Inna, A., Sendi, J., & Edy, M. 2010. Awar–Awar (Ficus Septica Burm. F.) Leaves Ethanolic Extract Induced Apoptosis Of Mcf-7 Cells By Downregulation of Bcl-2. Majalah Obat Tradisional. Vol 15(3): 100-104.
Sivanesan, D., Azad, H., & Rathinavelu, A. (2015). Apoptosis Induction by Ocimum sanctum Extract in LNCaP Prostate Cancer Cells. Journal of Medicinal Food. Vol. 18 (7) : 1–10
Sliwkal, L., Wiktorskal, K., Suchocki, P., Lizwa, P., Jaromin, A., Flis, A., & Chilmonczyk, Z. (2016). The Comparison of MTT and CVS Assays for the Assessment of Anticancer Agent Interactions. Plosone.
Sukardiman, Ekasari, W., & Hapsari, P. (2006). Anticancer and Induction if Apoptosis Activity of Chloroform Fraction From Daun Pepaya (Carica papaya L) in Myeloma Cancer Cells Culture. Media Kedokteran Hewan; Vol.22(2):104-111.
Teissier, S., Pang, C.L., & Thierry, F. (2010). The E2F5 repressor is an activator of E6/E7 transcription and of the S-phase entry in HPV18-associated cells. Oncogene; 29(36): 5061-70.
Vidhya, N. & Niranjali, D.S. (2011). Induction of Apoptosis By Eugenol in Human Breast Cancer Cells. Indian Journal of Experimental Biology; 49 (11): 871–878.
Wikanta, T., Rasyidin, M., Rahayu, L., Pratitis, A. (2012). Cytotoxic Activity and Apoptosis Induction of Ulva fasciata Delile Ethyl Acetate Extract Against CaSki and MCF-7 Cell Lines. JP Perikanan ;7(2): 87–96.
Wu, S.Y., Leu, Y.L., Chang, Y.L., Wu, T.S., Kuo, P.C., & Liao. (2012). Physalin F Induces Cell Apoptosis in Human Renal Carcinoma Cells by Targeting NF-kappaB and Generating Reactive Oxygen Species. Plos ONE; 7: e40727.
Wzeerapreeyakul, N., Nonpunya, A., Barusrux, S., Thitimetharoch, T. and Sripanidkulchai, B. (2012). Evaluation of the anticancer potential of six herbs against a hepatomacellline. J. Chinese Medicine. 7(15).